The first-line pharmacological treatment for major depressive disorder (MDD) are antidepressant drugs known as selective serotonin re-uptake inhibitors (SSRIs). But a significant proportion of people don’t respond to these drugs.
Given that major depression is a global mental health problem that is on the increase, it is important to find novel pharmacological treatments for those who do not respond to the current ones. But to do that, we need to understand exactly how the drugs work – which we currently don’t.
MDD is a debilitating and distressing mental health disorder, trapping sufferers in a rigid and negative state of mind. There’s even evidence suggesting that this lack of flexibility is associated with cognitive changes, including negative thoughts and biases, and problems with learning and memory.
In our new study, published in Molecular Psychiatry, we show that an SSRI called ecitalopram may actually make brains more “plastic” – meaning more flexible and adaptive; more able to facilitate communication between neurons (brain cells). Brain plasticity is simply the ability of neural circuits to change through growth and reorganisation. Learning involves brain plasticity, including changes in neural circuits, and can help people to recover from depression.
One novel treatment option for depression, approved by the US Food and Drug Administration, is intranasal esketamine (an anaesthetic made from ketamine), although it has not as yet been approved for use by the NHS. The psychedelic drugs LSD and psilocybin are also being investigated for treatment resistant depression in research studies, but are not yet approved by regulatory bodies. When these studies are conducted, there is careful monitoring by a medical professional to ensure participant safety.
We know that both SSRIs and psychedelics target the same brain receptor (known as the 5HT-2A). By contrast, eskatamine, similar to ketamine, works on a different receptor (N-methyl-D-aspartate or NMDA) and affects the brain chemical glutamate.
So how do SSRIs and psychedelics work to reduce symptoms of depression? At present, we don’t have the full picture. But the 5HT-2A receptor is linked to the brain chemical serotonin, increasing levels of it in the brain. And a recent study has indeed shown that serotonin appears to be reduced in people with depression.
SSRIs, however, also affect the neurotransmitters GABA and glutamate. The latter has been linked to learning, cognition and memory – suggesting SSRI may actually help to restore cognitive function. Although the exact mechanisms of psychedelics are not yet fully understood, their antidepressant effects seem to work in a similar way to SSRIs given their effects on 5HT-2A receptors. However, there are also other reactions to psychedelics, such as hallucinations.
Measuring brain plasticity
All these drugs have therefore been suggested to affect brain plasticity. However, in humans, it can be difficult to estimate levels of brain plasticity. One common method that scientists have used is to measure a protein called the brain-derived neurotrophic factor (BDNF) in blood samples.
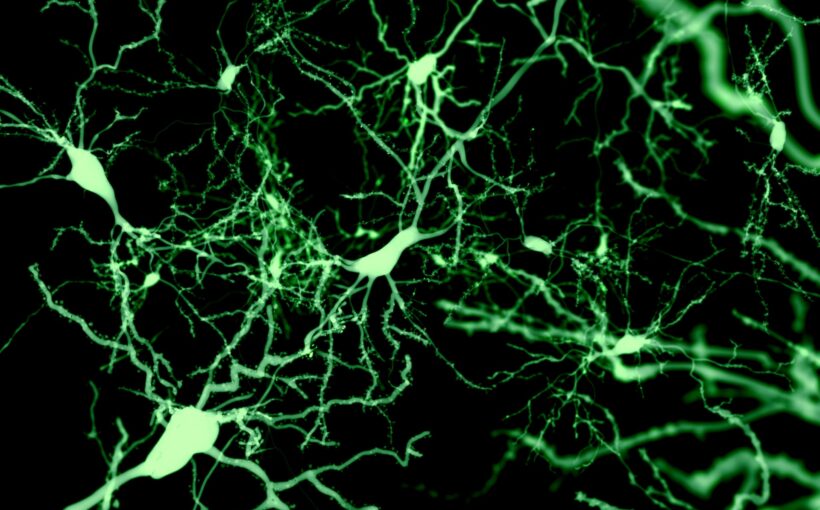
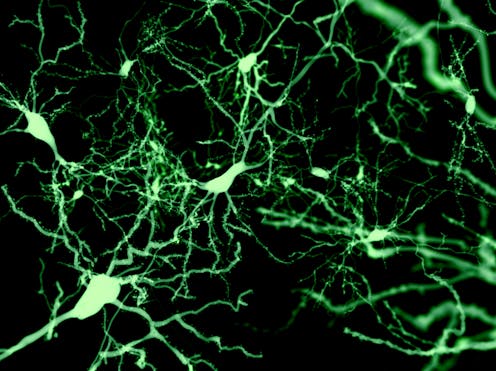
BDNF helps brain plasticity by increasing the number of synapses (locations where neurons can communicate with each other), as well as the branches and growth of developing neurons. Synapses are particularly important in brain functioning as they allow transmission of chemical and electrical signals from one neuron to another. Similarly, synapses also store brain chemicals for release.
There have been some studies showing that antidepressant drugs increase BDNF. However, better techniques are required to study plasticity in the human brain.

To develop better drugs, one approach is to find anti-depressant drugs with a faster mechanism of action. According to the NHS website, SSRIs usually need to be taken for two to four weeks before any benefit is felt.
We suspected that one reason for this delayed effect may be that brain plasticity needs to occur with SSRI treatment. As this process involves rewiring, such as the creation of synapses and circuits, it isn’t instant, but is thought to take approximately 14-21 days.
In our study, which was a collaboration between the University of Cambridge and the University of Copenhagen, we used a novel technique to measure plasticity in the human brain, following SSRI treatment, for the first time.
Thirty-two participants underwent positron emission tomography (PET) scanning to detect the amount of a protein called “synaptic vesicle glycoprotein 2A”, or SV2A, in the brain. We know that SV2A is a marker of the presence of synapses. An increased amount would suggest that more synapses are present and therefore that brain plasticity is higher.
Our results showed a rise in this protein as a result of taking escitalopram (an SSRI). We found that, in those taking escitalopram, increased SV2A was associated with increased duration on the drug. Our findings suggest that brain plasticity increases over three to five weeks in healthy humans following daily intake of escitalopram.
This is the first real evidence in humans that SSRIs really do boost neuroplasticity – seen in the brain – and that this is one of the reasons it can treat depression. Similar evidence from studies in the human brain are still required for the psychedelics.
It makes sense that if antidepressant treatment facilitates brain plasticity, this should make it easier for people taking these treatments to learn new things. And we know that the ability to adopt new strategies, and change them if they don’t work (supported by what researchers call cognitive flexibility), is key to recovering from depression.
![]()
Barbara Jacquelyn Sahakian receives funding from the Leverhulme Trust and the Lundbeck Foundation. Her research work is conducted within the NIHR Cambridge Biomedical Research Centre (BRC) Mental Health and Neurodegeneration Themes and the NIHR MedTech and in vitro diagnostic Co-operative (MIC). She consults for Cambridge Cognition.
Christelle Langley receives funding from the Leverhulme Trust. Her research work is conducted within the NIHR Cambridge Biomedical Research Centre (BRC) Mental Health and Neurodegeneration Themes and the NIHR MedTech and in vitro diagnostic Co-operative (MIC).